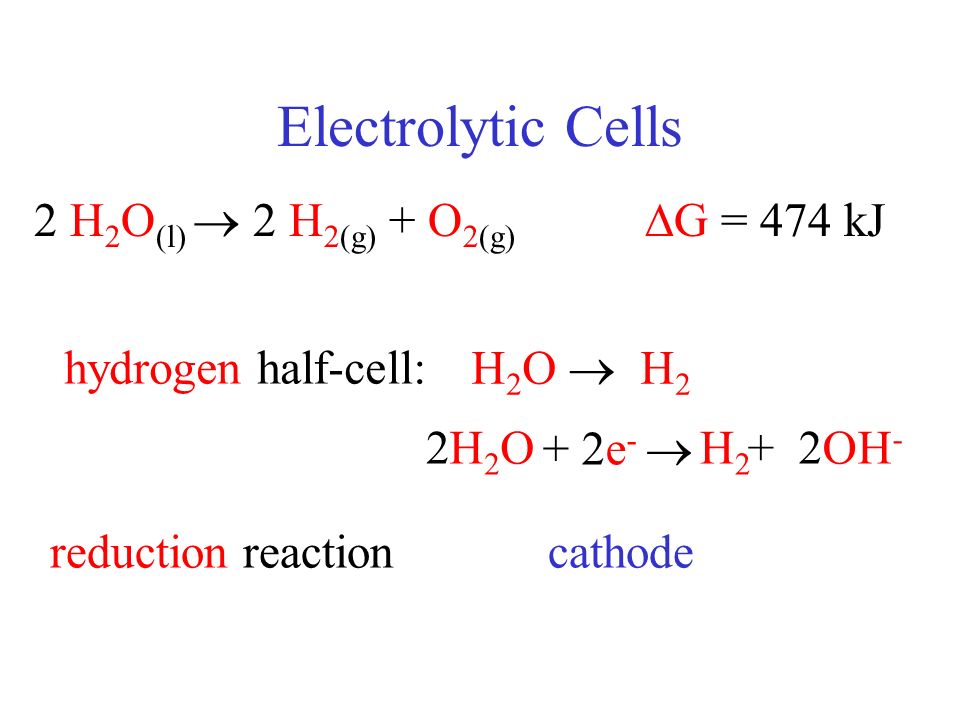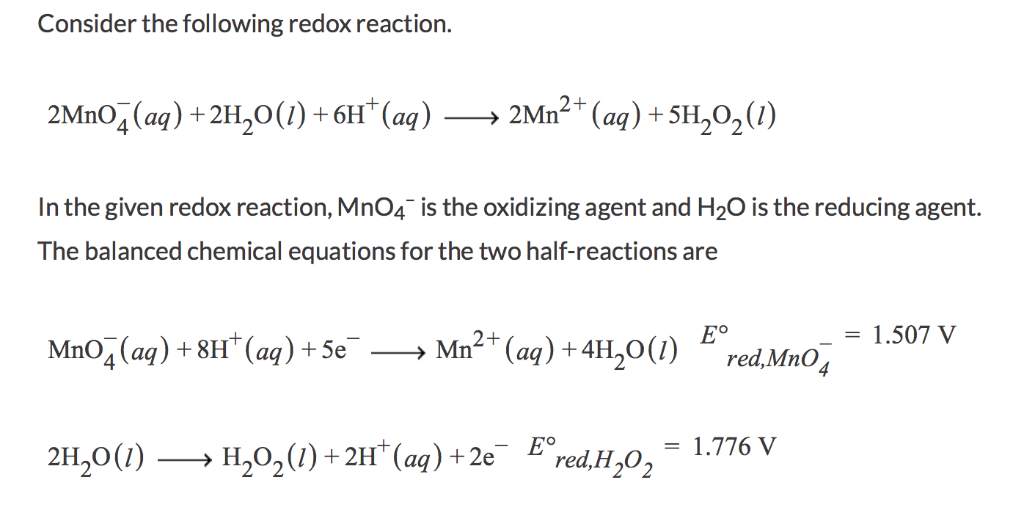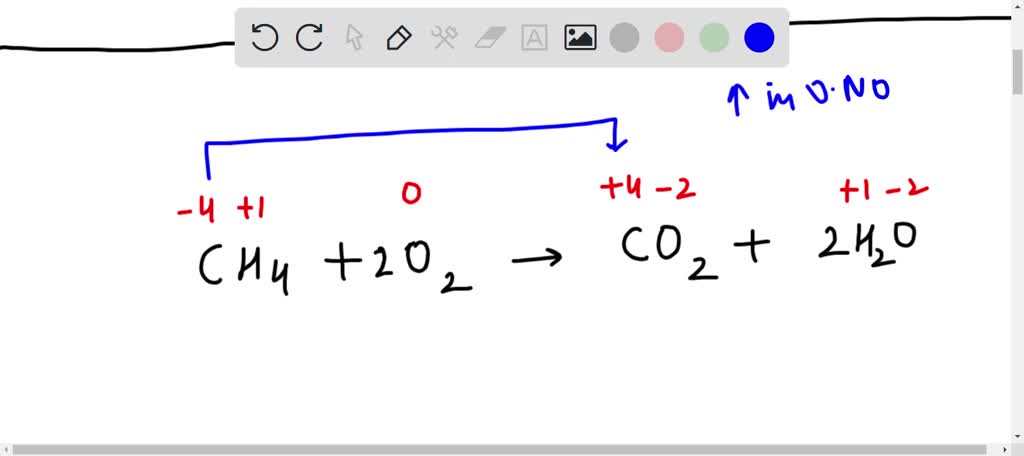
SOLVED: Which substance functions as a reducing agent in the following redox reaction? CH4 + 2 O2 → CO2 + 2H2O CH4 O2 CO2 H2O
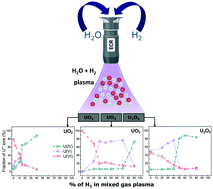
Mixed H2O/H2 plasma-induced redox reactions of thin uranium oxide films under UHV conditions - Dalton Transactions (RSC Publishing)

For the redox reaction, MnO^ - 4 + C2O^2 - 4 + H^ + → Mn^2 + + CO2 + H2O , the correct coefficients of the reactants for the balanced equation are:
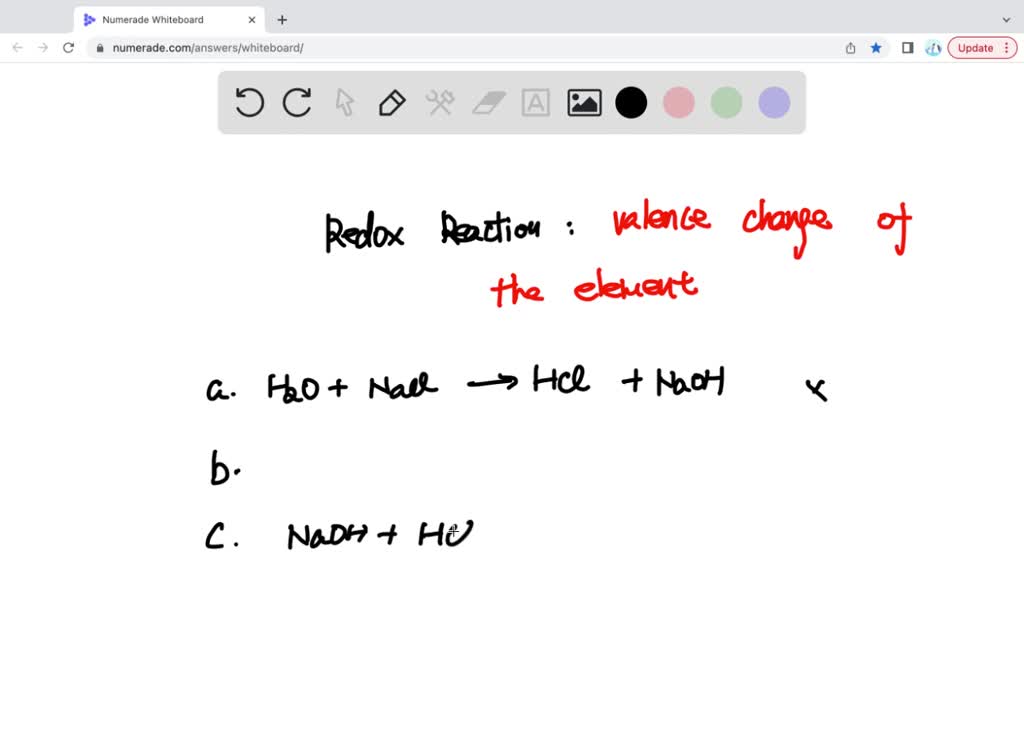
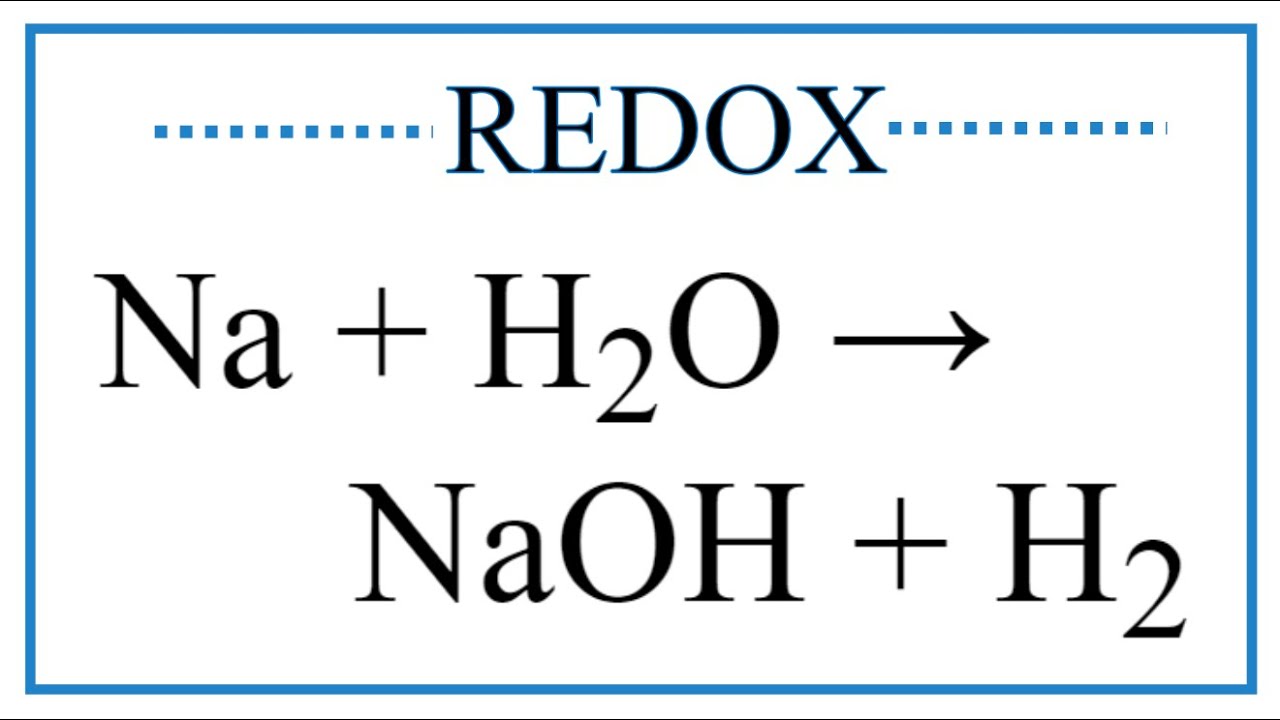
SOLVED: Which one of the following reactions is a redox reaction? a H2O +NaCl → NaOH + HCl b None of these reactions are redox. c NaOH + HCl → NaCl +

SOLVED: Balance the following redox reaction in acidic solution by inputting the correct coefficients. Fe2+ + O2 → Fe3+ + H2O If H+ or H2O is not present as reactant or product,