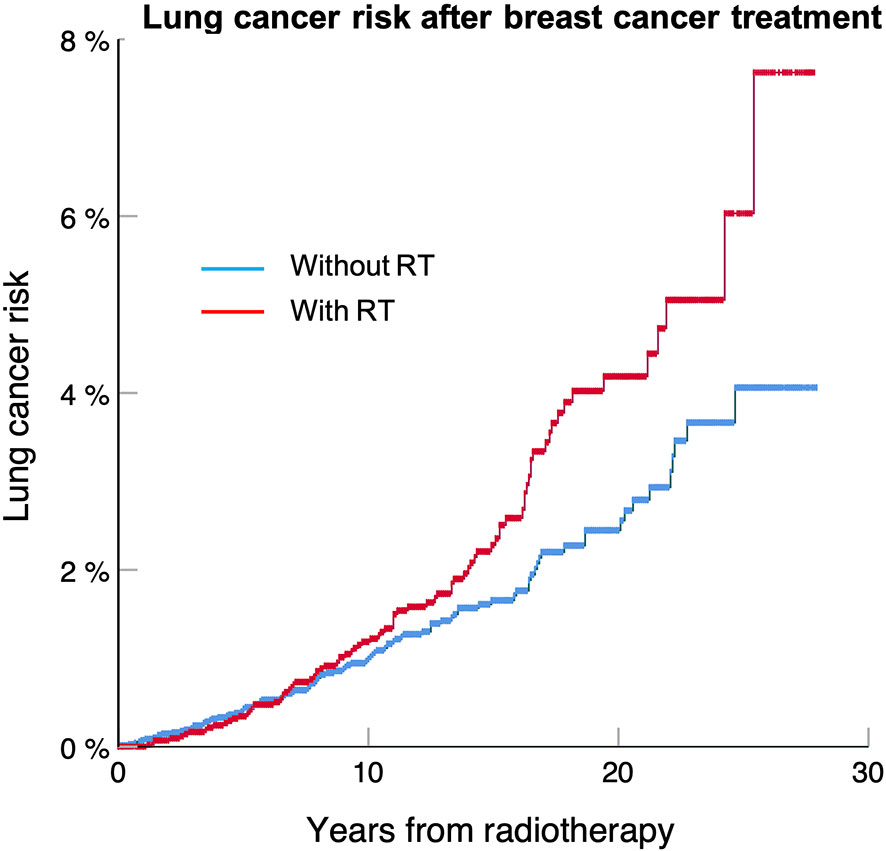
Frontiers | Estimation of Annual Secondary Lung Cancer Deaths Using Various Adjuvant Breast Radiotherapy Techniques for Early-Stage Cancers

Development of flexible electrochemical impedance spectroscopy-based biosensing platform for rapid screening of SARS-CoV-2 inhibitors - ScienceDirect

PDF) Heck Reaction Catalyzed by Nanosized Palladium on Chitosan in Ionic Liquids | Nicoletta Ditaranto - Academia.edu

Palladium-catalyzed highly regioselective Buchwald-Hartwig amination of 5-substituted-1,2,3-triiodobenzene: Facile synthesis of 2,3-diiodinated N-arylanilines as potential anti-inflammatory candidates - ScienceDirect

Pharmaceuticals | Free Full-Text | New Derivatives of the Multi-Stage Active Malaria Box Compound MMV030666 and Their Antiplasmodial Potencies

PDF) Catalytic hydrogenation of nitrile rubber using palladium and ruthenium complexes | Roberto Silva de Souza - Academia.edu

Cancers | Free Full-Text | Feasibility Study on the Radiation Dose by Radioactive Magnetic Core-Shell Nanoparticles for Open-Source Brachytherapy












