Organic-NaH2PO4-H2O aqueous biphasic system for extraction of paeonol from cortex moutan: Solvent selection and mechanism probing - ScienceDirect

10049-21-5, 137.99, Sodium Phosphate, Monobasic, Monohydrate, Crystal, Reagent, ACS - 6NNX7|S1395-2.5KG - Grainger
Welcome to Chem Zipper.com......: In a determination of P an aqueous solution of NaH2PO4 is treated with a mixture of Ammonium and magnesium ions to precipitate magnesium Ammonium phosphate (Mg(NH4)PO4.6H2O. This is

El monohidrato de fosfato de sodio Dihyrogen Nah2PO4· H2O. - China Mono fosfato de sodio, Metal detergente

Mono fosfato de sodio el tratamiento de agua Productos químicos Nah2PO4. H2O. - China Fosfato monosódico, Dihidrógeno fosfato de sodio
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%201KG.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $36.00 : Bioland Scientific, for Your Research Needs

Phase Equilibrium for the Ternary System KH2PO4 + NaH2PO4 + H2O at 303.15 K | Journal of Chemical & Engineering Data

1 Bán Hóa chất Sodium dihydrogen phosphate monohydrat, reagent grade - NaH2PO4.H2O - SO0331 - Scharlau giá rẻ ở hcm

10049-21-5, 137.99, Sodium Phosphate, Monobasic, Monohydrate, Crystal, Reagent, ACS - 6NNX9|S1395-500GM - Grainger
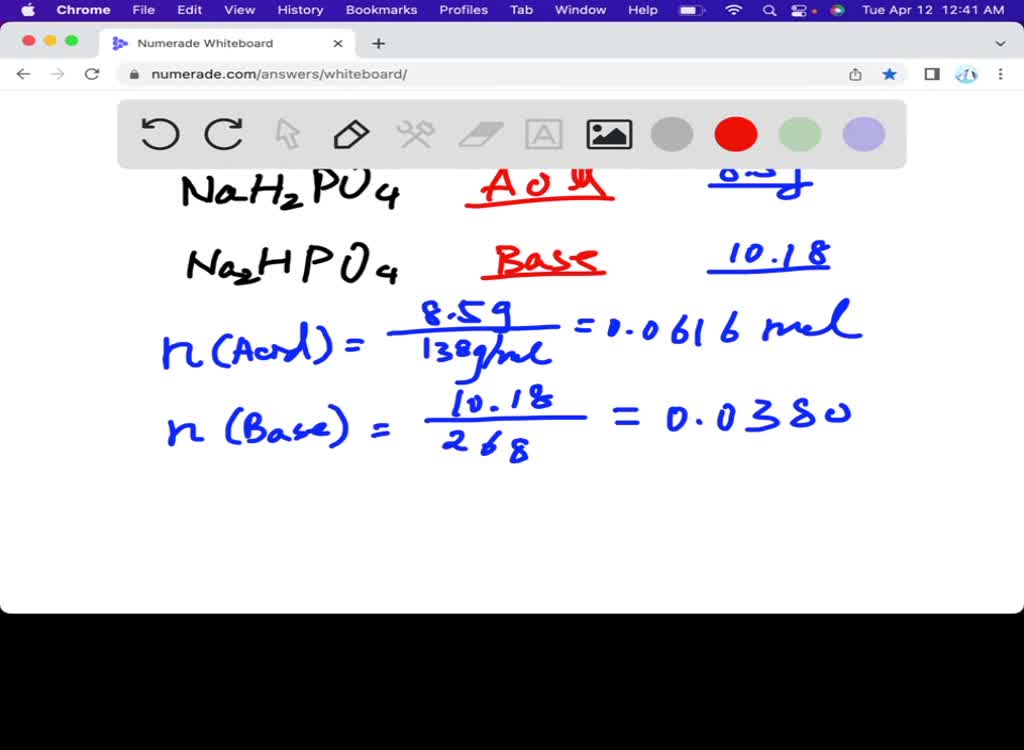
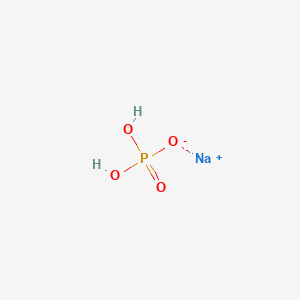
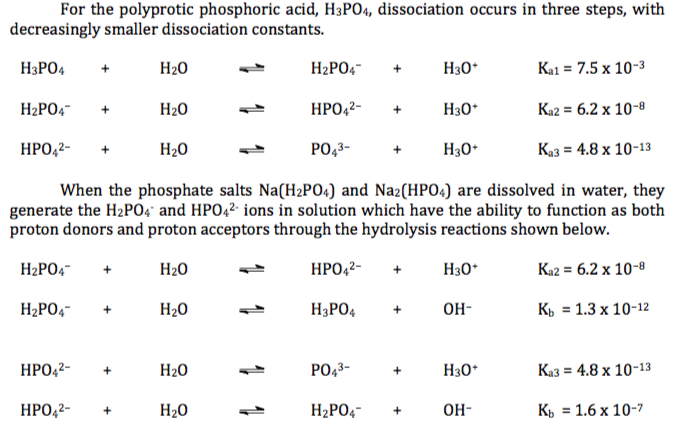
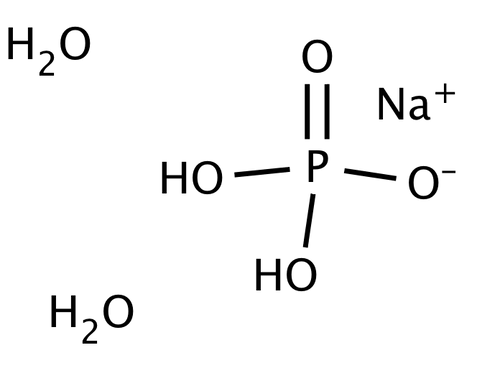
SOLVED: 1. How many g of Na2HPO4 and NaH2PO4 2H2O would you need to prepare 1L of 0.1M sodium phosphate buffer pH 7.0? (Hint= use the Henderson-Hasselbalch equation) Express your answer to