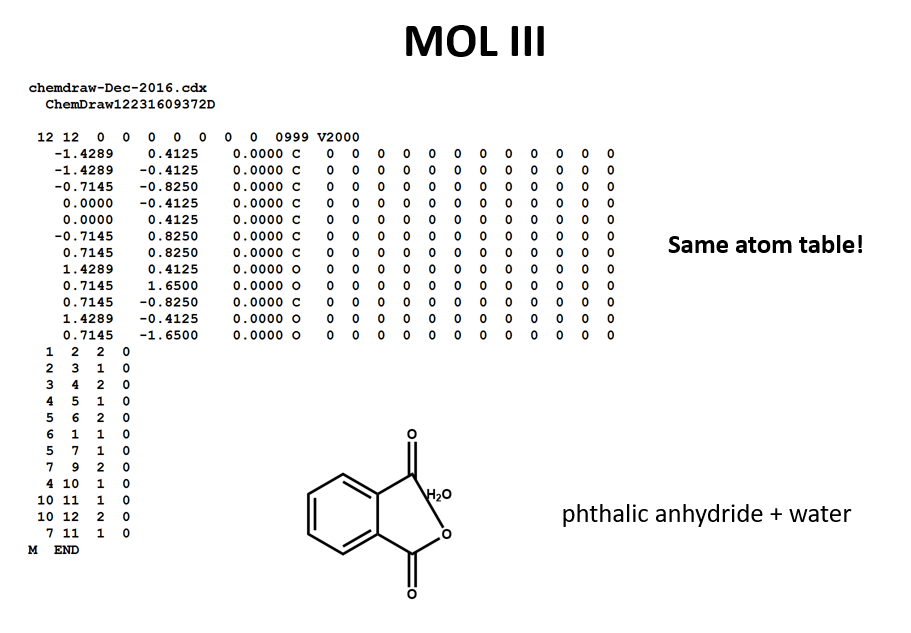
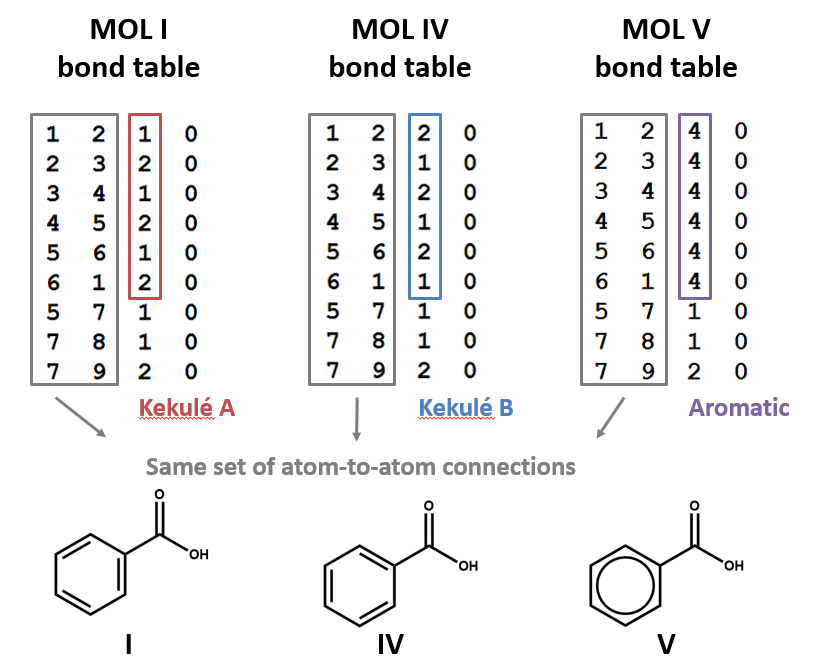
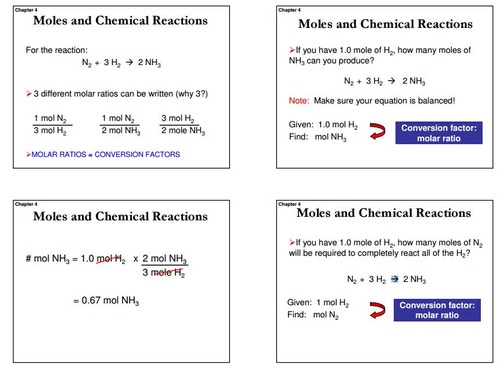
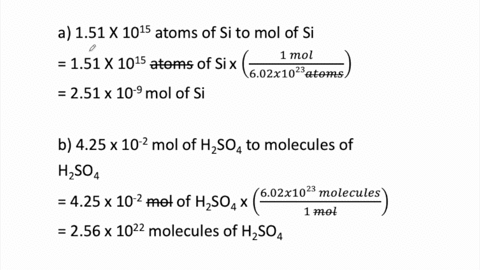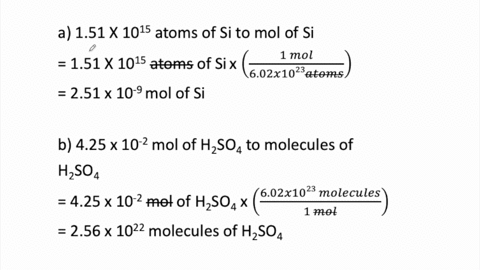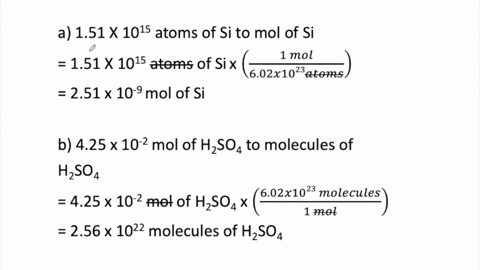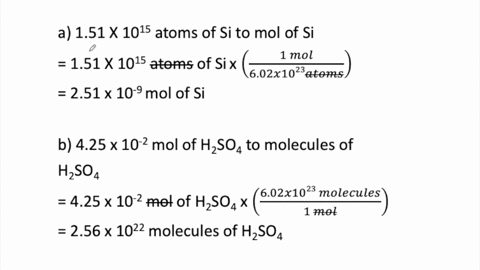STOICHIOMETRY CALCULATIONS COACH COX. MOLE TO MOLE CONVERSIONS Converting from moles of one substance in a chemical reaction to moles of another substance. - ppt download
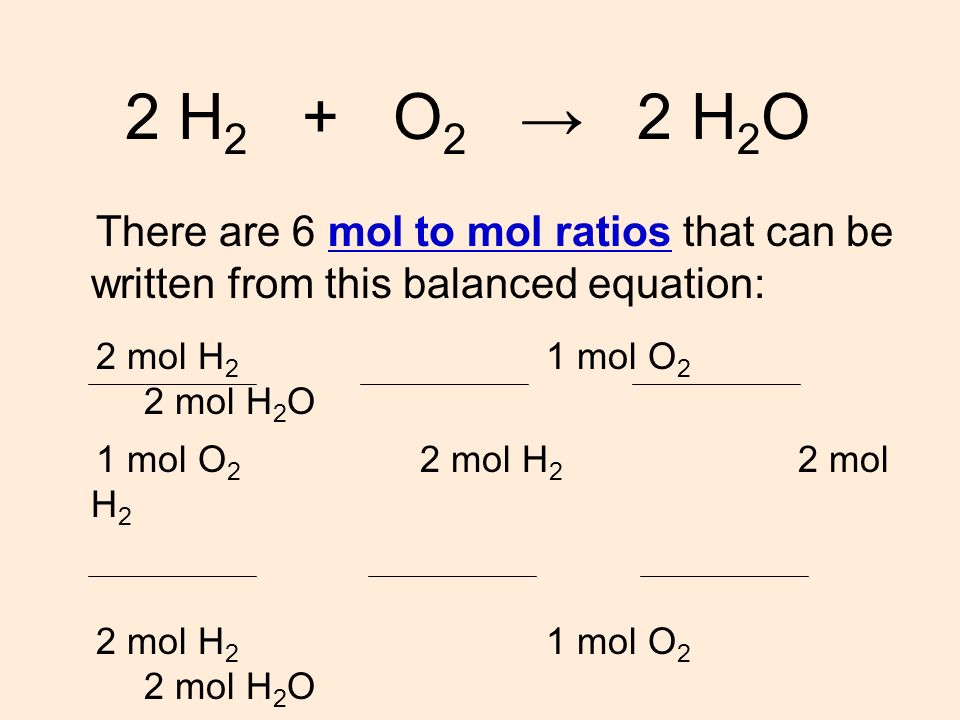
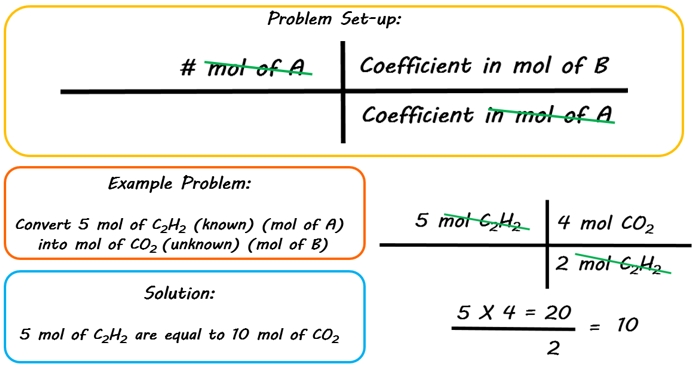
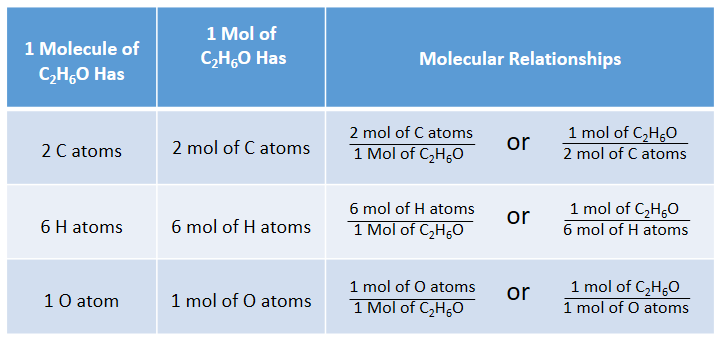
Mol ratio: coefficients of a balanced equation 2 H 2 + O 2 → 2 H 2 O 2 mol H 2 for every 1 mol O 2 In chemical calculations, mol ratios convert moles of. - ppt download
2 moles of an ideal gas at temp 27 degree c is heated isoermall from volume v to 4v .if R=2 cal/mol then the heat input in the process is approximately

For one mole of a van der Waals' gas when b = 0 and T = 300 K , the PV vs 1/V plot is shown below. The value of the van

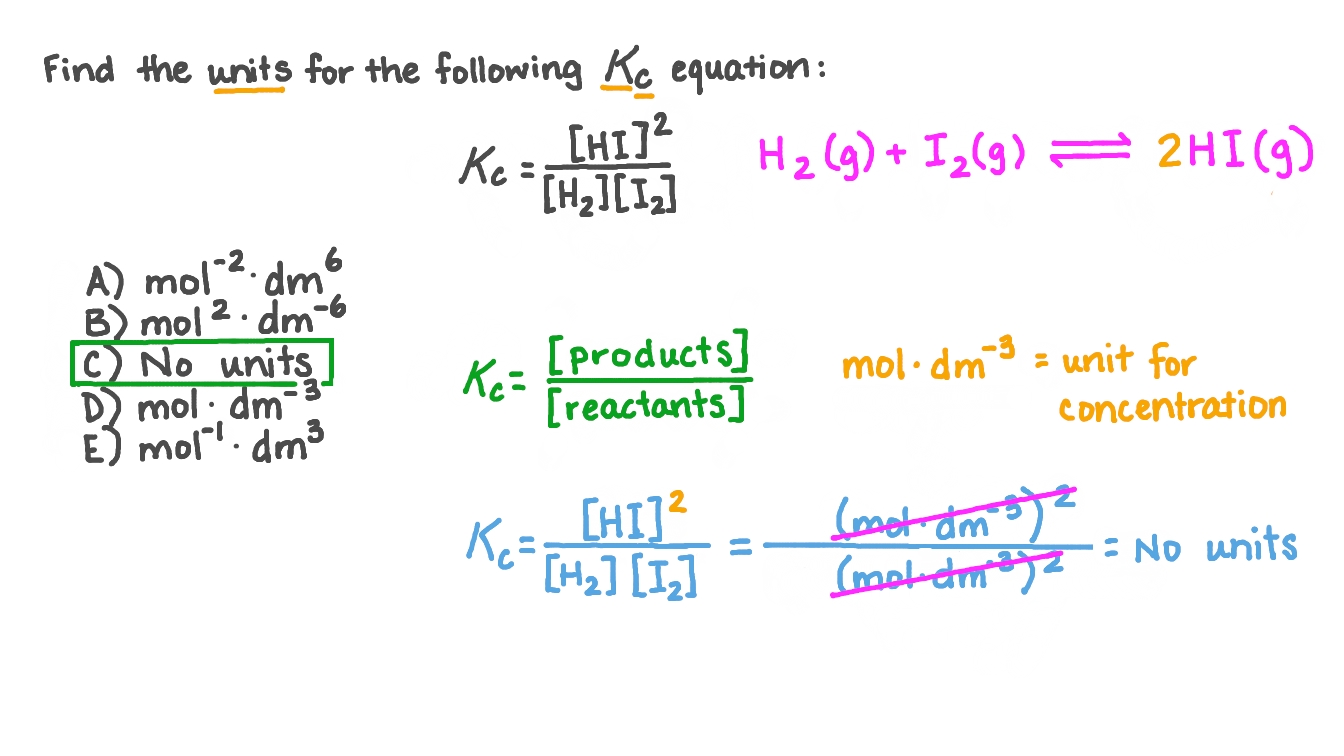
The rate constant of a reaction is 9.2 × 10 5 mol 2 L 2 s 1. The order of the reaction is:A. 3B. 2C. ZeroD. 1