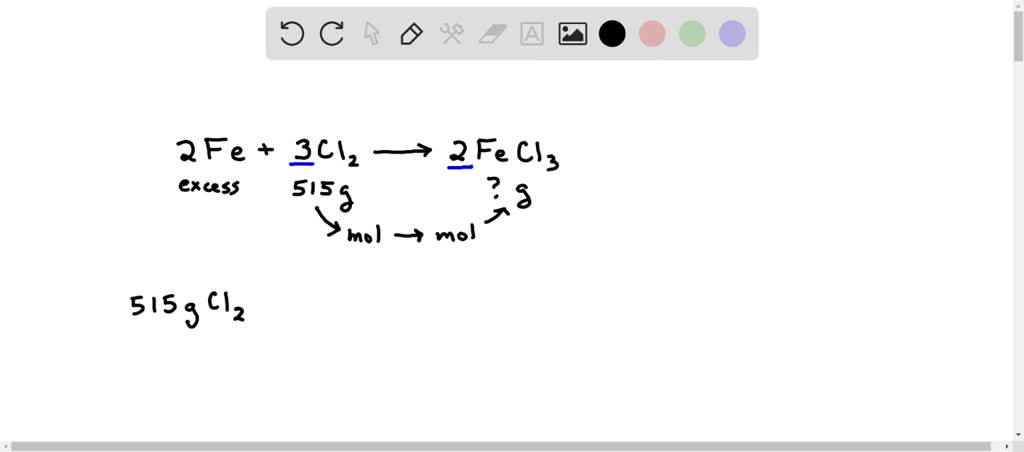
SOLVED: Iron metal and chlorine gas react according to this unbalanced chemical equation: Fe(s) + Cl2(g) ⟶ FeCl3(s) How many grams of FeCl3 are produced when 515 g Cl2 react with excess

Miami, Florida, USA - September 14, 2019: Hyundai Santa Fe Car on Shell Gas Station in Miami USA Editorial Image - Image of fuel, expensive: 161231130