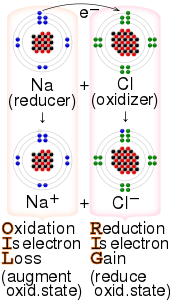
Which of the following would have a noble gas configuration; Na^(+2), O^-3, Cl^-1, or Ca^(+3)? Why? | Socratic
![SOLVED: Click on the electron pair that will become pi electrons and allow the regeneration of the aromatic ring :Ci= :-AI-Gi: CI H CH3 CH3 CH3 Rechedk e (12 of 13] 7th SOLVED: Click on the electron pair that will become pi electrons and allow the regeneration of the aromatic ring :Ci= :-AI-Gi: CI H CH3 CH3 CH3 Rechedk e (12 of 13] 7th](https://cdn.numerade.com/ask_images/14070eb0ea7847ba85ead2cf8cb2781c.jpg)
SOLVED: Click on the electron pair that will become pi electrons and allow the regeneration of the aromatic ring :Ci= :-AI-Gi: CI H CH3 CH3 CH3 Rechedk e (12 of 13] 7th

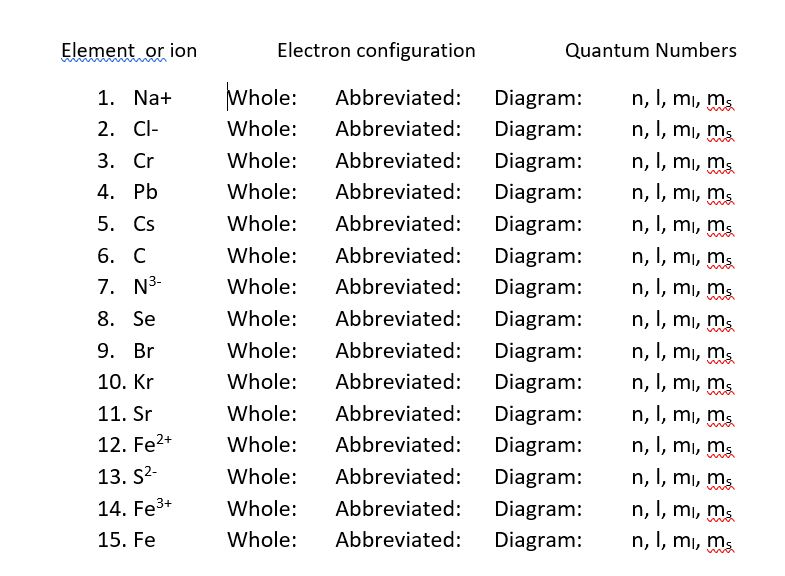
Write the electron configuration for the following: (CI, Zn, Ca^2+, F^-) a. Write one type of chemical bonds and one type of chemical reactions example for everyone? b. Write Lewis structure for

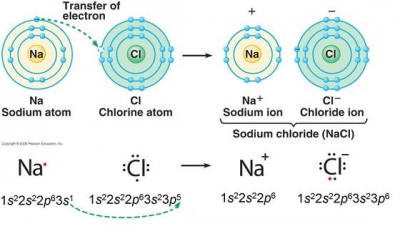
The diagram shows the electron configuration around the chlorine nucleus. Chlorine has 17 protons. CI Which - Brainly.com