Selective CO2 capture through microporous Tb(BTC)(H2O).(DMF)1.1 MOF as an additive in novel MMMs fabricated from Matrimid® 5218 - ScienceDirect

SOLVED: With the help of the activity series Of metals, write the products of the reactions und balance the equations. HNOs b) Ag HzSO4 Mg HCI Mg Mgo Balance the above equation;
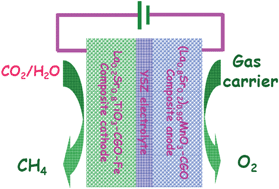
Direct synthesis of methane from CO2/H2O in an oxygen-ion conducting solid oxide electrolyser - Energy & Environmental Science (RSC Publishing)

SOLVED: Thiophene is carbon-hydrogen-sulfur compound used in the manufacture of pharmaceuticals: When burned completcly in excess oxygen; its combustion products ie COz, H,O, und SOz Combustion of a 0.535-8 sample yields 4.19

Mechanisms of CoII and Acid Jointly Catalyzed Domino Conversion of CO2, H2, and CH3OH to Dialkoxymethane: A DFT Study | ACS Catalysis

Formation of Formic Acid, Formaldehyde, and Carbon Dioxide by Electron-Induced Chemistry in Ices of Water and Carbon Monoxide | ACS Earth and Space Chemistry

Syngas and Synfuels from H2O and CO2: Current Status - Nguyen - 2015 - Chemie Ingenieur Technik - Wiley Online Library

CO2-H2O mixtures in the geological sequestration of CO2. I. Assessment and calculation of mutual solubilities from 12 to 100°C and up to 600 bar - ScienceDirect

Carbon Dioxide as a C1 Building Block for the Formation of Carboxylic Acids by Formal Catalytic Hydrocarboxylation - Ostapowicz - 2013 - Angewandte Chemie International Edition - Wiley Online Library