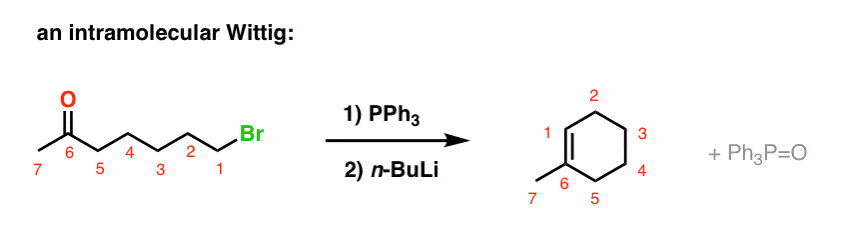
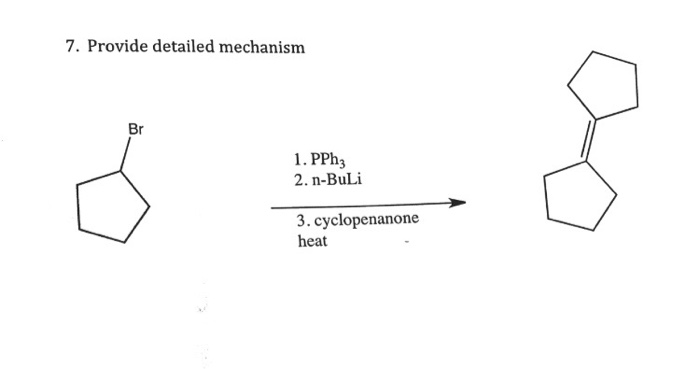
Scheme 4. Reagents and conditions: (a) n-BuPPh 3 Br, n-BuLi, THF, 0 °C;... | Download Scientific Diagram

Scheme 2. (a) Boc-N 3 , Et 3 N, dioxane, rt (90%); (b) s-BuLi, TMSCl,... | Download Scientific Diagram
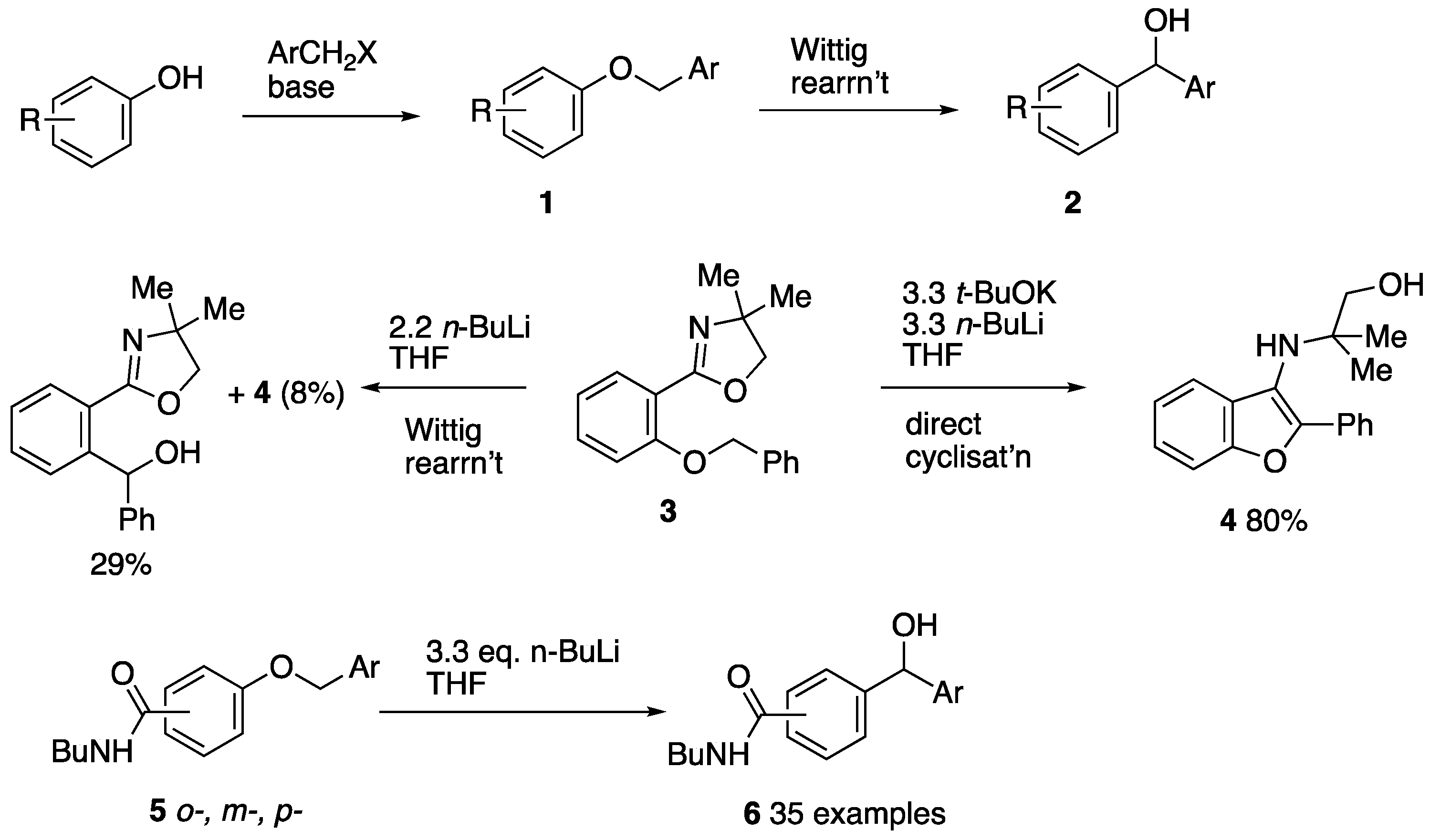
Organics | Free Full-Text | Synthesis and Wittig Rearrangement of 3- and 4-Benzyloxyphenylphosphonamidates

Scheme 16 i) CrO 3-Pyr; ii) n-BuLi, 3-furylmethyl triphenylphosphorane;... | Download Scientific Diagram

Crystal Structures of n-BuLi Adducts with (R,R)-TMCDA and the Consequences for the Deprotonation of Benzene | Journal of the American Chemical Society

Thiophilic ring opening reactions of 3,3-bis(trifluoromethyl)-5-alkoxy-1,2-dithiolanes under action of nucleophiles - ScienceDirect
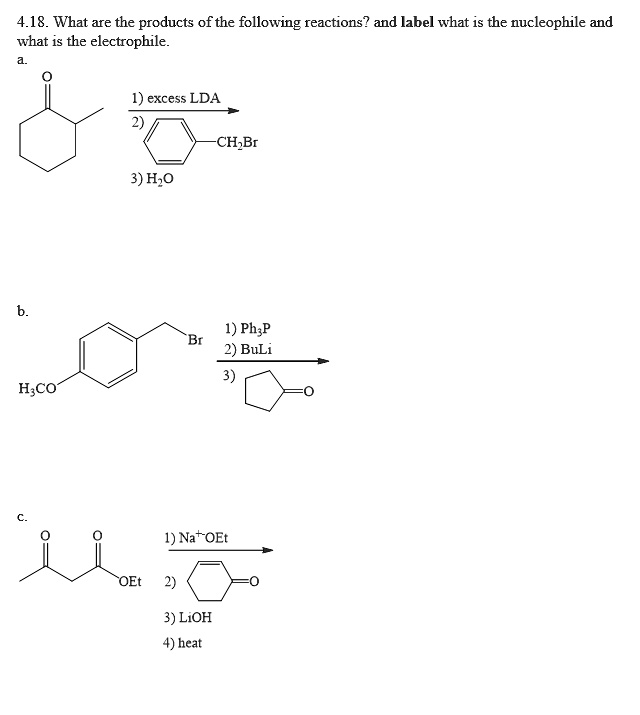
SOLVED: 4.18 What are the products of the following reactions? and label what is the nucleophile and what 1s the electrophile excess LDA CHBr 3) H,o 1) Ph;P 2) BuLi H;cO 1)
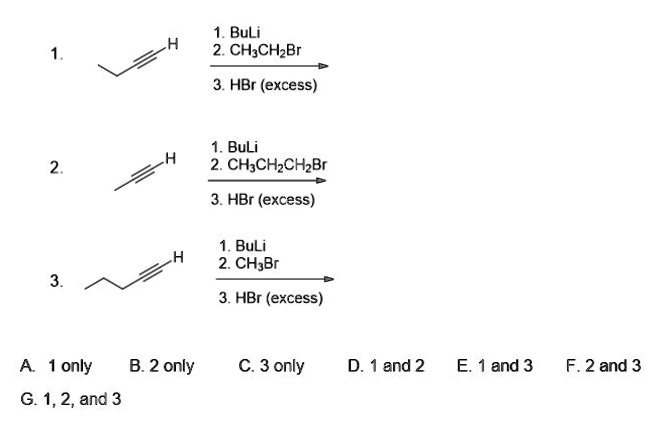
SOLVED: Buli 2 CH;CH Br 3 HBr (excess) BuLi 2. CH;CHzCHzBr 3. HBr (excess) BuLi 2. CH;Br 3. HBr (excess) 1 only B. 2 only 6. 1,2, and 3 C.3 only and 2 and 3 F.2and 3

Asymmetric Deprotonation using s-BuLi or i-PrLi and Chiral Diamines in THF: The Diamine Matters | Journal of the American Chemical Society





![Answered: [1] Ph3P Br a. [2] BuLI H. [3] b. [1]… | bartleby Answered: [1] Ph3P Br a. [2] BuLI H. [3] b. [1]… | bartleby](https://content.bartleby.com/qna-images/question/e910c6f7-542c-4c5c-b26b-e166f57f7d0f/a75ed780-74cc-4e40-9c1c-5bedacaffd1f/hagx0x.png)