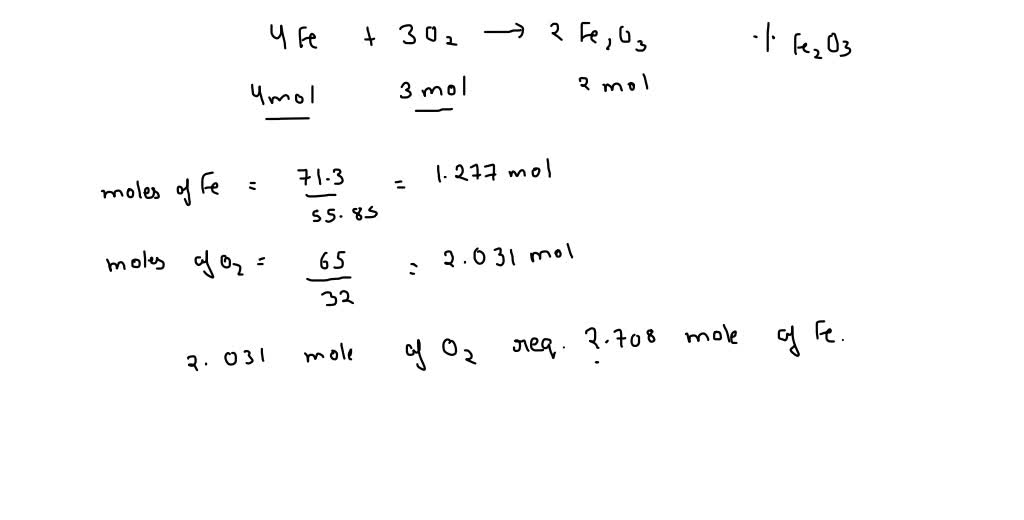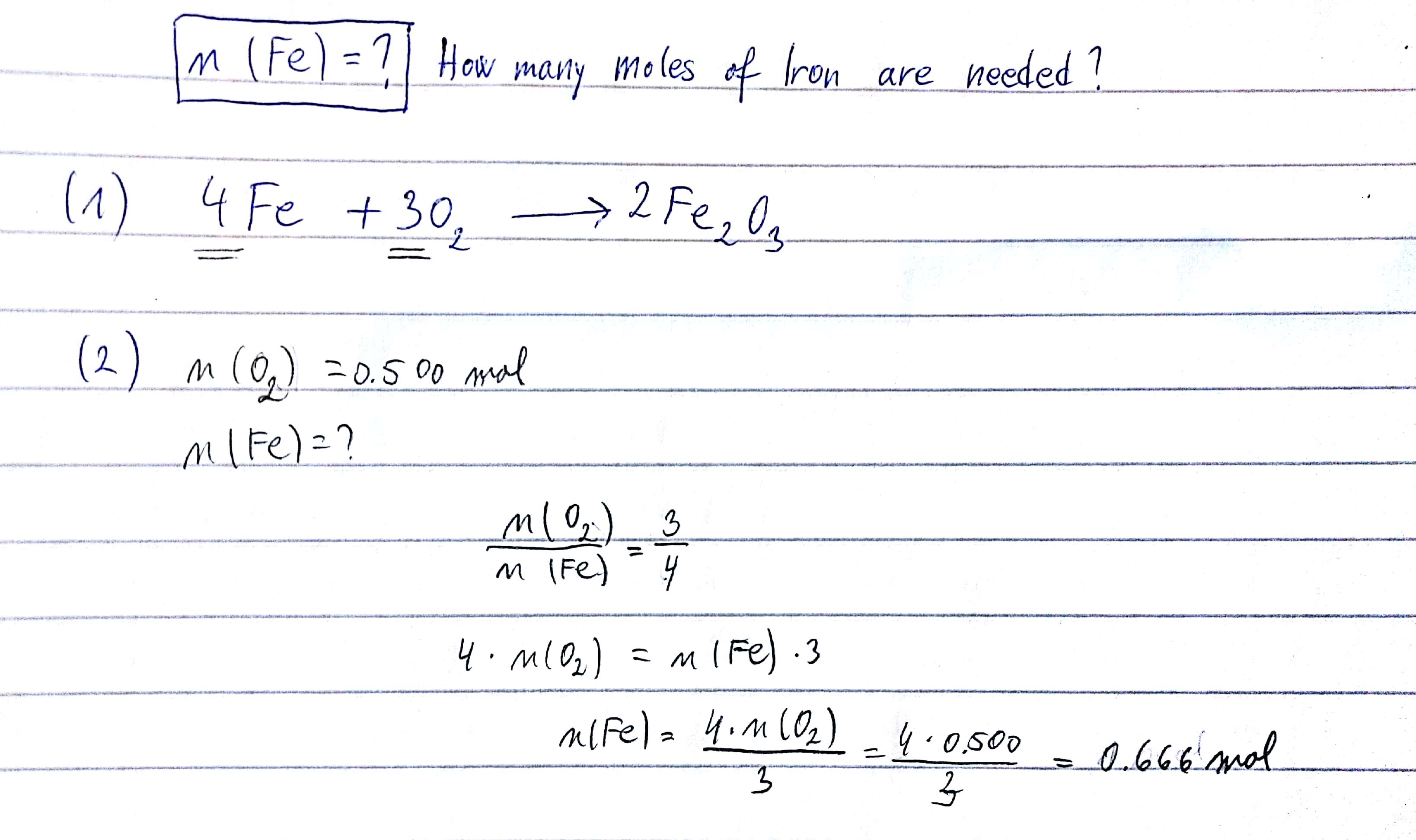SOLVED: Consider the following reaction: 4Fe (s) + 3O2 (g) —> 2Fe2O3 (s) If 5.0 moles of Fe and 4.0 moles of O2 are the present as the reactants, determine the limiting

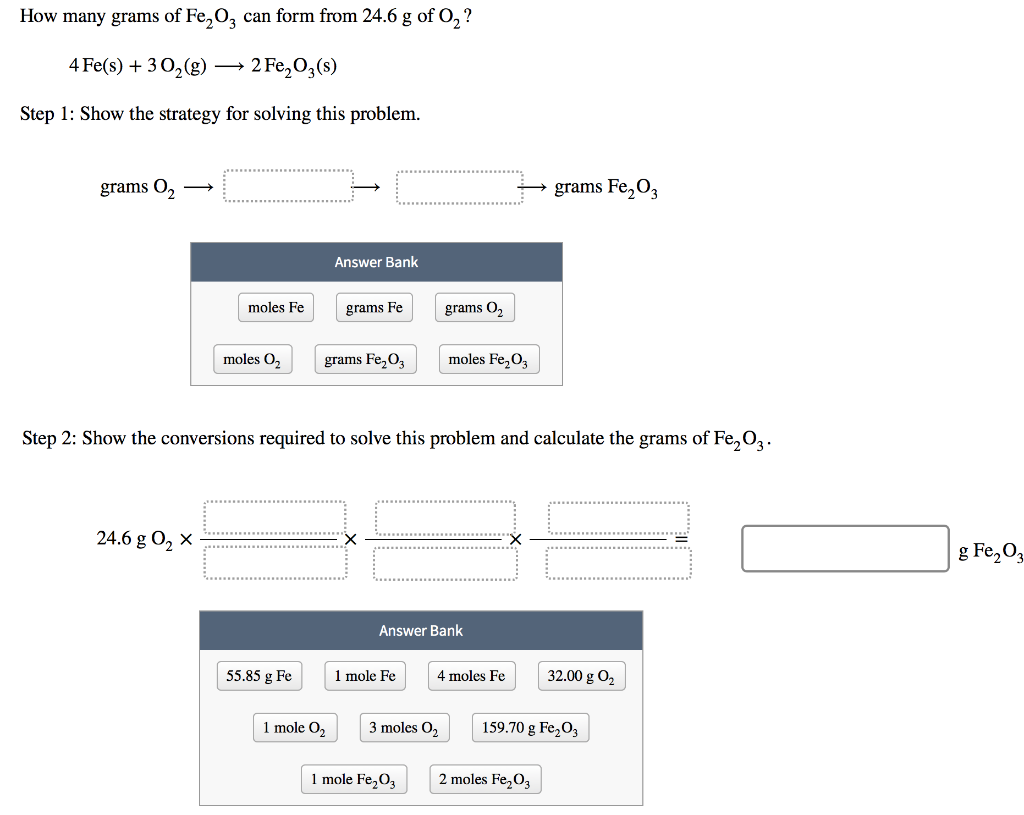
SOLVED: 4Fe + 3O2 2Fe2O3 If 44.2 grams of oxygen react with an excess of iron, what mass of Fe2O3 can form? (Fe2O3= 159.70 g/mol) a. 147 g b. 331 g c. 294 g d. 221 g