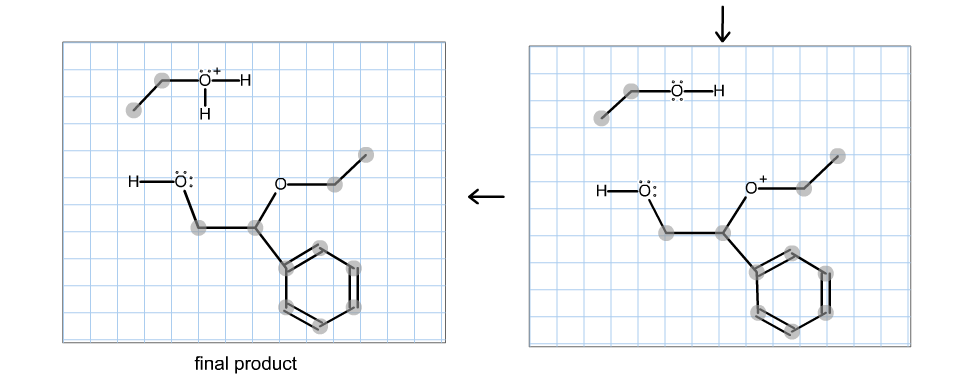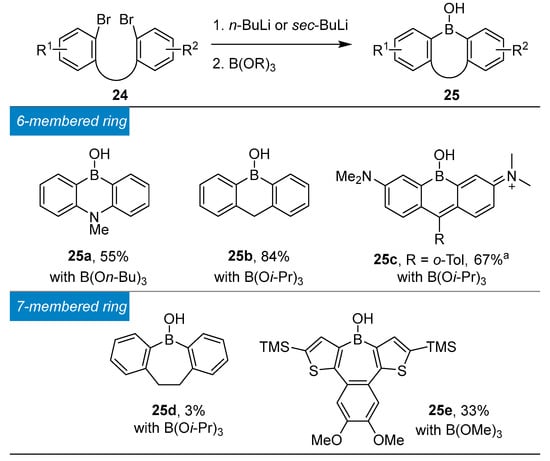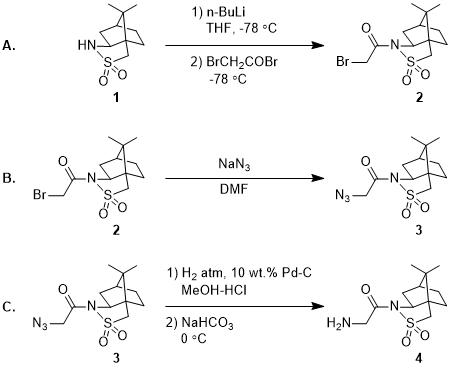
Molecules | Free Full-Text | Aziridine Ring Opening as Regio- and Stereoselective Access to C-Glycosyl-Aminoethyl Sulfide Derivatives

NMR and DFT Studies with a Doubly Labelled 15N/6Li S‐Trifluoromethyl Sulfoximine Reveal Why a Directed ortho‐Lithiation Requires an Excess of n‐ BuLi - Hédouin - Angewandte Chemie International Edition - Wiley Online Library

Metalation of Pyridines with nBuLi−Li−Aminoalkoxide Mixed Aggregates: The Origin of Chemoselectivity | Journal of the American Chemical Society

NMR and DFT Studies with a Doubly Labelled 15N/6Li S‐Trifluoromethyl Sulfoximine Reveal Why a Directed ortho‐Lithiation Requires an Excess of n‐ BuLi - Hédouin - Angewandte Chemie International Edition - Wiley Online Library
![Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances](https://www.science.org/cms/asset/860963e6-85a5-48b3-9e47-4a596c258578/abd5290-f1.gif)
Lewis acid–catalyzed domino generation/[2,3]-sigmatropic rearrangement of ammonium ylides to access chiral azabicycles | Science Advances

Are n-BuLi/TMEDA-Mediated Arene Ortholithiations Directed? Substituent-Dependent Rates, Substituent-Independent Mechanisms | Journal of the American Chemical Society


![Stats] 2. Bundesliga table after matchday 25. : r/soccer Stats] 2. Bundesliga table after matchday 25. : r/soccer](https://preview.redd.it/7h0vyfk96xl81.jpg?auto=webp&s=ef20eb5c5e005905b30e2903b65f90e8ae894f6c)
![Solved OZO Br NiBry, Ir[dFppy]z(dtbby)PF6 CsHCO3, BlueLED | Chegg.com Solved OZO Br NiBry, Ir[dFppy]z(dtbby)PF6 CsHCO3, BlueLED | Chegg.com](https://media.cheggcdn.com/media/ed5/ed57a899-fd4a-4095-93ea-6e74405e49f3/phpU6uXgx.png)